Bringing It Home: How Ab Biotechnology Successfully In-Housed Recombinant Protein Production
At Ab Biotechnology, we’re always excited to take on new challenges so we were thrilled when a potential client came to us with a transformed E. coli BL21 (DE3) strain for expression of a recombinant protein as part of a wider, overarching agreement.
Historically we have tended to outsource bespoke manufacture of recombinant proteins, however managing this remotely can bring its own challenges. To our minds, it would be advantageous to bring this project in-house as we would be better able to control timelines, quality attributes and costs. Initially, we needed to ask ourselves two important questions…………. Can we do it? And is it a commercially viable project; is it cheaper to outsource?
The starting point was to look at staff CVs and establish what experience we have working on recombinant proteins; it turns out we had many years of experience within our Development team in recombinant protein expression using E. coli systems and this was complemented by well-established protein purification expertise. So, there was no need to recruit, we just needed to dust off some old skills and establish the expert team to steer the project!
These are the key questions we asked ourselves;
• 𝗗𝗼 𝘄𝗲 𝗿𝗲𝗮𝗹𝗹𝘆 𝘂𝗻𝗱𝗲𝗿𝘀𝘁𝗮𝗻𝗱 𝘁𝗵𝗲 𝗺𝗲𝘁𝗵𝗼𝗱𝗼𝗹𝗼𝗴𝘆?
• 𝗪𝗵𝗮𝘁 𝘄𝗼𝘂𝗹𝗱 𝗶𝘁 𝘁𝗮𝗸𝗲 𝘁𝗼 𝘀𝘂𝗰𝗰𝗲𝘀𝘀𝗳𝘂𝗹𝗹𝘆 𝗯𝗿𝗶𝗻𝗴 𝘁𝗵𝗲 𝗻𝗲𝘄 𝗽𝗿𝗼𝗰𝗲𝘀𝘀 𝗶𝗻-𝗵𝗼𝘂𝘀𝗲?
That meant asking a lot of practical questions:
• 𝗗𝗼 𝘄𝗲 𝗵𝗮𝘃𝗲 𝘁𝗵𝗲 𝗿𝗶𝗴𝗵𝘁 𝗲𝗾𝘂𝗶𝗽𝗺𝗲𝗻𝘁?
• 𝗔𝗿𝗲 𝘁𝗵𝗲 𝗻𝗲𝗰𝗲𝘀𝘀𝗮𝗿𝘆 𝗿𝗲𝗮𝗴𝗲𝗻𝘁𝘀 𝗮𝗻𝗱 𝗰𝗼𝗻𝘀𝘂𝗺𝗮𝗯𝗹𝗲𝘀 𝗮𝘃𝗮𝗶𝗹𝗮𝗯𝗹𝗲?
• 𝗛𝗼𝘄 𝗹𝗼𝗻𝗴 𝗱𝗼𝗲𝘀 𝗲𝗮𝗰𝗵 𝘀𝘁𝗮𝗴𝗲 𝘁𝗮𝗸𝗲, 𝗮𝗻𝗱 𝗵𝗼𝘄 𝗱𝗼𝗲𝘀 𝘁𝗵𝗮𝘁 𝗳𝗶𝘁 𝘄𝗶𝘁𝗵𝗶𝗻 𝗼𝘂𝗿 𝗽𝗿𝗲-𝗲𝘅𝗶𝘀𝘁𝗶𝗻𝗴 𝘄𝗼𝗿𝗸𝗳𝗹𝗼𝘄𝘀?
It wasn’t just about following a protocol; it was about making sure everything would run smoothly within our own setup. We knew some tweaks would be needed, but that’s part of what we enjoy—taking on a challenge, understanding the details, making it work in our environment and hopefully using our expertise to add improvements as we progress.
Once we had satisfied our own questions we went back to the client with a proposal to make the recombinant protein in-house, with timelines and costs. We returned with a signed contract!
A contract is all well and good, however the success and failure of many projects comes down to defining the key project deliverables upfront. Here we produced two overarching documents:
• 𝗣𝗿𝗼𝗷𝗲𝗰𝘁 𝗦𝗰𝗼𝗽𝗲 - 𝗱𝗲𝗳𝗶𝗻𝗶𝗻𝗴 𝘄𝗵𝗮𝘁 𝗮𝗿𝗲 𝘁𝗵𝗲 𝗽𝗿𝗼𝗷𝗲𝗰𝘁 𝗱𝗲𝗹𝗶𝘃𝗲𝗿𝗮𝗯𝗹𝗲𝘀, 𝘁𝗵𝗲 𝗾𝘂𝗮𝗹𝗶𝘁𝘆 𝗮𝘁𝘁𝗿𝗶𝗯𝘂𝘁𝗲𝘀, 𝗵𝗼𝘄 𝘄𝗶𝗹𝗹 𝘁𝗵𝗲 𝗽𝗿𝗼𝗷𝗲𝗰𝘁 𝗯𝗲 𝗺𝗮𝗻𝗮𝗴𝗲𝗱 𝗮𝗻𝗱 𝗲𝘀𝘁𝗮𝗯𝗹𝗶𝘀𝗵𝗶𝗻𝗴 𝗺𝗲𝗰𝗵𝗮𝗻𝗶𝘀𝗺𝘀 𝘁𝗼 𝗺𝗼𝗻𝗶𝘁𝗼𝗿.
• 𝗣𝗿𝗼𝗷𝗲𝗰𝘁 𝗚𝗮𝗻𝘁𝘁 - 𝗲𝘀𝘁𝗮𝗯𝗹𝗶𝘀𝗵𝗶𝗻𝗴 𝗮 𝘁𝗶𝗺𝗲𝗹𝗶𝗻𝗲, 𝗿𝗲𝘀𝗼𝘂𝗿𝗰𝗲 𝗮𝘃𝗮𝗶𝗹𝗮𝗯𝗶𝗹𝗶𝘁𝘆 𝗮𝗻𝗱 𝗶𝗱𝗲𝗻𝘁𝗶𝗳𝗶𝗰𝗮𝘁𝗶𝗼𝗻 𝗼𝗳 𝗰𝗿𝗶𝘁𝗶𝗰𝗮𝗹 𝗽𝗮𝘁𝗵𝘀.
Both documents were signed by the client and Ab Biotechnology so that we had clear agreement on the project goals.
Weekly project meetings were then set up across all stake holder departments to monitor project progress and to ensure no project creep or change of scope occurred.
Using preliminary data provided by our client, protocols were established for all critical process steps such as cell cultivation, inclusion body isolation, protein refolding and purification.
We were fortunate to be able to drive costs down by using some pre-existing equipment for the project, but the team were excited to purchase some shiny new items such as an autoclave, shaking incubator and freeze dryer. URS documents were written, the equipment arrived, and qualification was performed. Now the work could begin!
E. coli is the simplest organism for expressing recombinant proteins. The system used extensively is based upon a plasmid engineered with a very tightly regulated promoter to allow different levels of protein expression depending on the concentration of inducer IPTG. The plasmid is also antibiotic-resistant ensuring that selection of the plasmid is maintained even at very high cell densities. There can be various problems when expressing proteins in E. coli and several schemes can be established to help overcome these problems. The most frequent issue encountered is the production of insoluble recombinant protein and having a method from our client for solubilisation and re-folding of the protein was a major advantage for the production process.
Laboratory work began with the E. coli expression method, and we were able to follow the client’s method for protein expression. We didn’t have a huge amount of detail for expression analysis, so we used our previous experience and knowledge to develop a method for analysis of soluble and insoluble protein expression which demonstrated that the protein was mostly insoluble as expected.
So far so good, but any protein scientist will tell you that this is where the challenges really begin since there are a multitude of different refolding techniques, buffers and additives that can be employed. Luckily, we had a good starting point which was the method information from our client, and we were able to adapt this to our lab setting. Some process steps theoretically required weekend input and working in the small hours of the morning, but our previous experience allowed us to tweak timings to suit our schedules in the knowledge that there would be no impact on the protein product.
We were very encouraged to see a large amount of soluble protein material present after the refolding reaction was completed. With the process going in our favour, we quickly progressed to purification using two different ion exchange chromatography steps. The final purified product was concentrated, filtered and lyophilised before storing safely in a freezer.
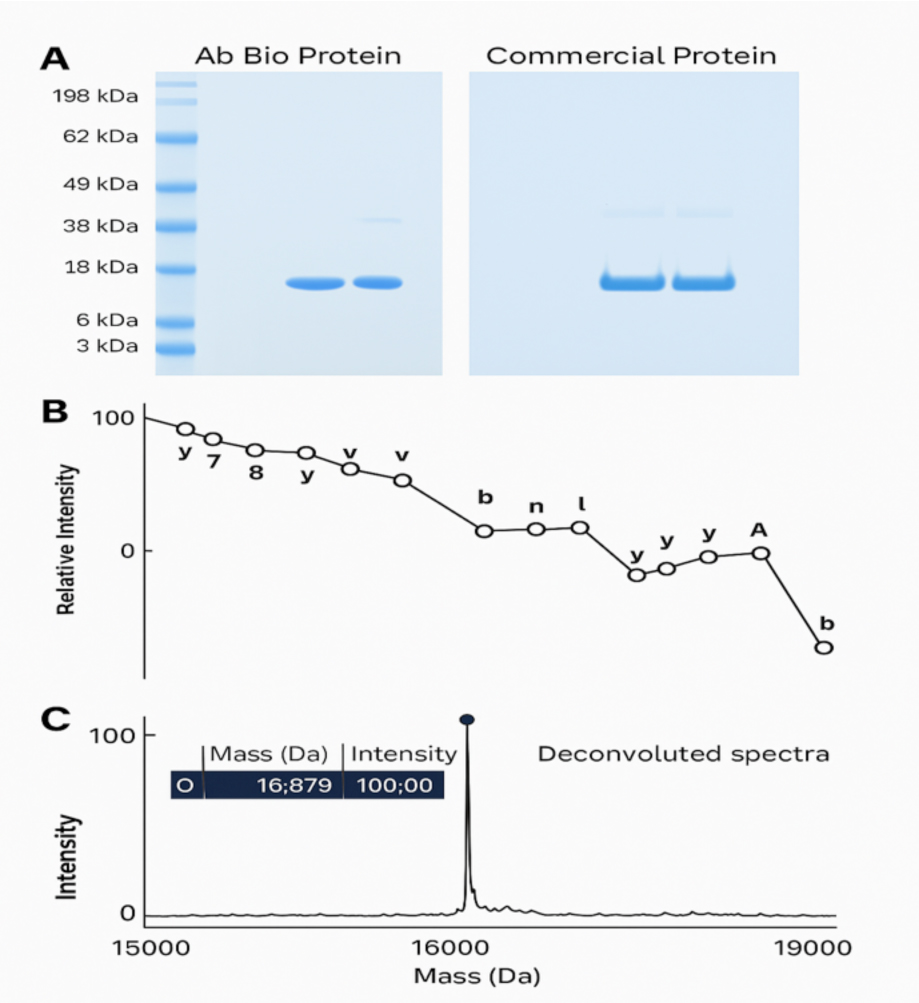
Now it was on to the analysis of the final product as the lyophilised protein was handed over to our Quality Control department. The Process Development team were confident in the quality of the product thanks to in-process testing performed during the production steps but would the finished product pass all the stringent QC testing, including N-terminal sequencing, mass spectrometry, SDS-PAGE, sterility, endotoxin, host cell protein and bioassay?
Of course it did! We were delighted to see that the purified product closely matched a commercially available (and much more expensive) product meeting all criteria for identity, purity/impurities and activity confirming the effectiveness of our in-house process. Our yield and production timelines were also in-line with expectations and thus our client’s requests were fulfilled. By utilising Ab Biotechnology’s expertise to bring this process in house we have gained significant strategic business and quality advantages; no longer having to rely on external bespoke manufacture, Ab Bio has gained the organisation better control over delivery timelines, product quality and significant cost savings that we able to pass onto our client!
Contact us today to discuss your Bio-Tech needs or request a quote: enquiries@abbiotechnology.com
Ab Biotechnology Ltd – Precision You Can Trust, Quality You Can Prove.

